An introduction to the semiconductor
Semiconductors are a unique class of materials with special electrical properties, they’re neither full conductors nor complete insulators. This in-between behavior allows us to control how current flows through them, especially when influenced by temperature, light, or the addition of certain elements (a process called doping). In technology, semiconductors are essential for both data processing and energy generation, solar panels being a prime example. One of the most groundbreaking inventions in this field was the transistor, a tiny component used for amplification, switching, and processing electrical signals. Without semiconductors, there would be no transistors, no chips, and ultimately, no modern electronics.
Let's Start with the Atom
The Building Blocks of Matter
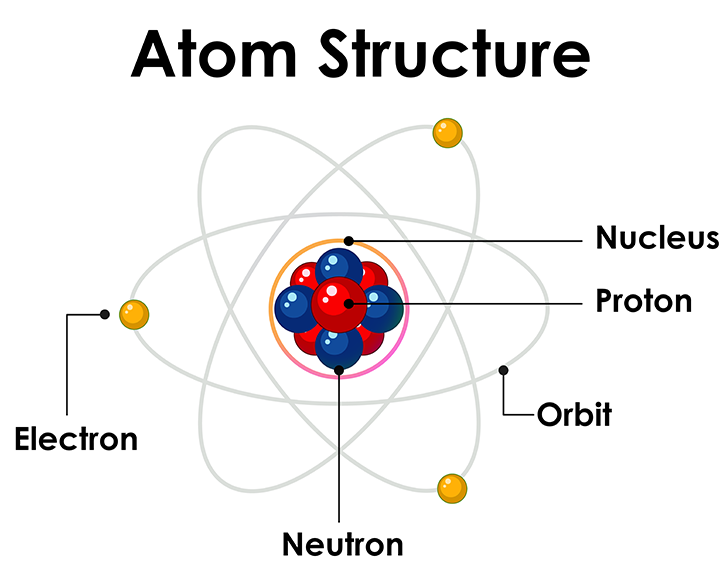
Everything around us (solids, liquids and gases) is made of atoms. Atoms are the basic units of matter and the building blocks of molecules. Each atom consists of three types of tiny particles: protons, neutrons, and electrons. Protons and neutrons are packed tightly together in the atom’s nucleus, at the center. Protons carry a positive charge, neutrons have no charge, and electrons (which are much lighter) orbit the nucleus and carry a negative charge. The number of protons defines the element and is equal to the number of electrons in a neutral atom. Neutrons help determine the mass of the atom. All known elements are organized in the periodic table, which groups them by their atomic number and shared properties.
Electrons and Energy Levels
From Einstein's Quantum Photon Theory
Electrons don’t just float randomly around the nucleus, they move in structured layers called shells or energy levels. These shells hold a set number of electrons, and the way these electrons are arranged plays a big role in how atoms interact. The outermost shell, known as the valence shell, is especially important because it determines how atoms bond or conduct electricity. In semiconductors, the behavior of electrons in these shells, especially how easily they can move between energy levels, is what makes them so useful in technology. For the sake of this article, we won’t dive into electron orbitals too deeply, because mostly the valence shell is what’s important for semiconductors.
Conductors, Insulators, and Semiconductors
What enables modern technology
To understand what makes semiconductors special, it helps to look at the two extremes: conductors and insulators. Conductors, like copper and silver, allow electricity to flow through them very easily. This is because their electrons, especially in the outer shell, are free to move. Insulators, on the other hand, like glass or rubber, hold on to their electrons tightly and hardly let any current pass through. Semiconductors sit right in the middle. Under the right conditions, they can behave like conductors, but they can also act like insulators when needed. This ability to switch between being an insulator and a conductor makes semiconductors incredibly useful. They can be turned “on” or “off,” allowing us to build complex circuits that respond to signals, store information, or convert energy — all at microscopic scales.
The Band Gap
A Key Concept
In solid-state physics, we often describe the movement of electrons using energy bands. The two most important bands are the valence band (where electrons usually are) and the conduction band (where electrons need to go to move freely and conduct electricity). The space between these two bands is called the band gap.
- Conductors - In conductors, the band gap is tiny or even overlapping — electrons move freely.
- Insulators - In insulators, the gap is large — electrons can’t jump across.
- Semiconductors - In semiconductors, the gap is just right. With a little energy — from heat, light, or electricity — electrons can make the jump. This makes the material switch from non-conductive to conductive when needed.
Doping Semiconductors
Customizing Conductivity
Pure semiconductors like silicon can already conduct electricity under the right conditions, but we can make them even more useful through a process called doping. This means adding tiny amounts of other elements to change how electrons behave.
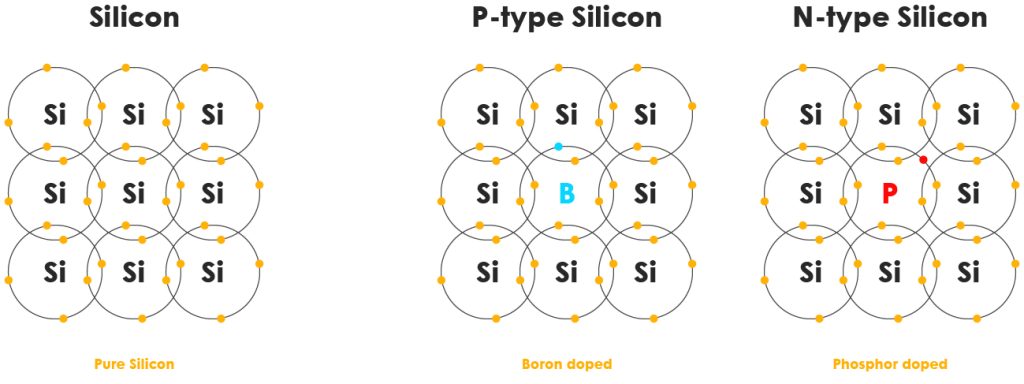
- N-type - If we add elements that have extra electrons, we get n-type semiconductors (n for negative). These extra electrons can move around easily.
- P-type - If we add elements that are missing electrons, we create p-type semiconductors (p for positive). These create “holes”, places where an electron is missing, which allows current to flow as electrons jump in to fill the gaps. Doping lets us control exactly how a semiconductor behaves, turning it into a highly flexible material for circuits.
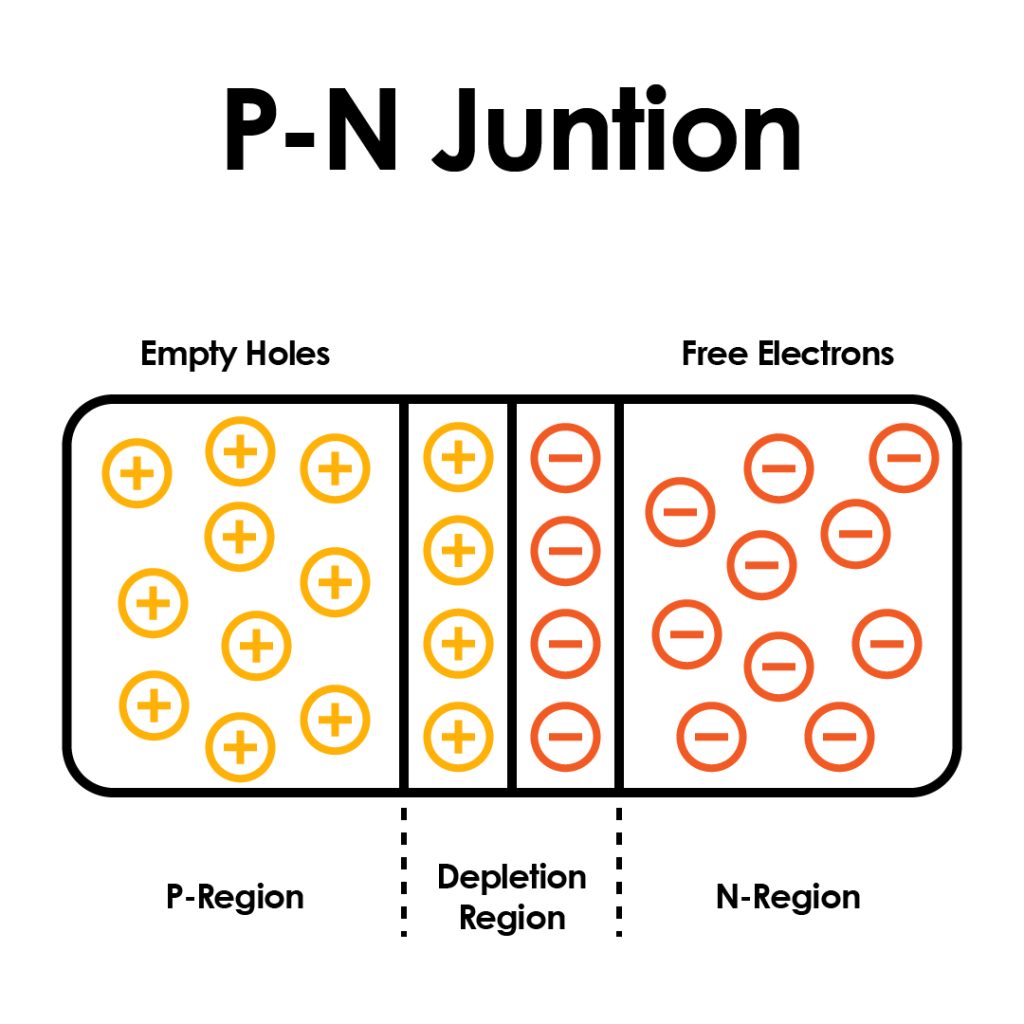
The PN Junction – The Heart of Semiconductor Devices
When a piece of p-type semiconductor is placed next to an n-type, we get a PN junction. This boundary between the two types creates a powerful effect: a built-in electric field. Electrons from the n-side are drawn to the holes on the p-side, but only under certain conditions can current flow through this junction. This makes it behave like a one-way valve for electricity, the basis for diodes and solar cells.
The PN junction is the core of many semiconductor devices. It’s how we can control when and how electricity moves, an essential function in any electronic system.

The Transistor – A Tiny Powerhouse
Take two PN junctions and place them together, and you get a transistor. This tiny component can act as a switch or an amplifier, depending on how it’s used. In a switch role, it can turn current on or off, just like a light switch, but at lightning-fast speeds. In amplification mode, it takes a small input signal and makes it much stronger.
Transistors are the building blocks of modern electronics. Your phone, laptop, and even your washing machine contain billions of them, all made from semiconductor materials. They process signals, store data, and allow complex computations to happen in the blink of an eye.
Photovoltaics – Turning Light into Electricity
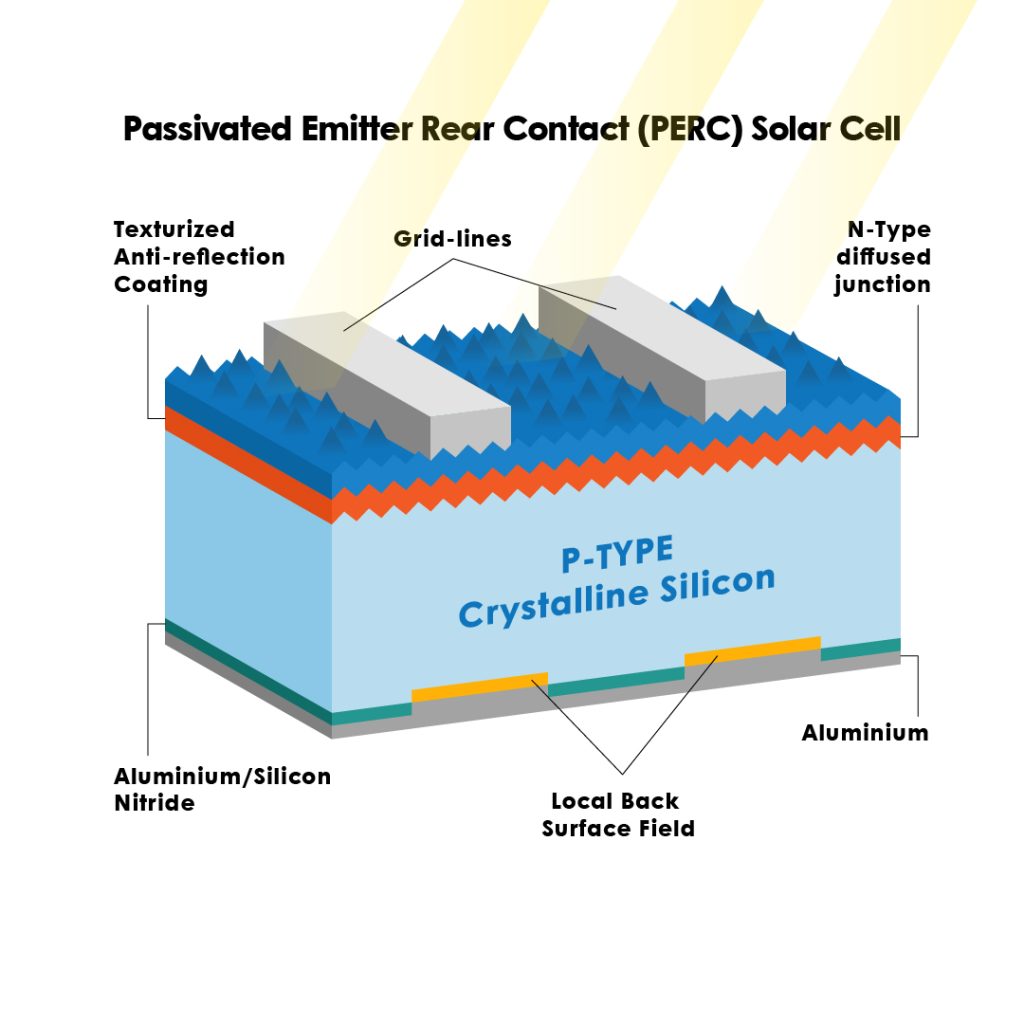
Semiconductors don’t just work with electricity, they can also interact with light. In solar cells, light particles (called photons) hit the surface of a semiconductor like silicon. If the energy of the photon is strong enough, it can knock an electron across the band gap into the conduction band, creating an electric current.
This is known as the photovoltaic effect. The structure of the solar cell, including a PN junction, then channels the movement of electrons to generate usable power. Without semiconductors, this clean, renewable energy technology wouldn’t exist.

Why Semiconductors Matter
From smartphones to solar panels, from medical devices to satellites, semiconductors are everywhere. Their ability to control electricity at the tiniest scale has revolutionized our world. Understanding the basics of atoms, electrons, and how semiconductors work helps us appreciate the incredible technology behind even the simplest gadgets. Whether you’re interested in renewable energy, computing, or electronics, semiconductors are at the core.